Utilizing resources normally reserved for English classes
I wrote a chapter on Balancing Equations and it can hopefully be found here: http://www.bookrix.com/_ebook-melissa-getz-balancing-equations/
The software would not recognize subscripts, much like Weebly doesn't, so the text is not ideal. I copied and pasted it here so you can read what I wrote here in case something goes goofy at the websites where I put the chapter on chemistry content.
After I wrote the student assignment, I went to issuu to republish this book. If that worked, then it should be embedded here.
The software would not recognize subscripts, much like Weebly doesn't, so the text is not ideal. I copied and pasted it here so you can read what I wrote here in case something goes goofy at the websites where I put the chapter on chemistry content.
After I wrote the student assignment, I went to issuu to republish this book. If that worked, then it should be embedded here.
My eBook: Balancing Equations
Why would we want to balance equations? Is there really a point to it? Well, it turns out that nature likes things to be organized, even though things spontaneously fall apart. Chemistry is full of what often feels like contradictions. You add an electron, the ion becomes more negative. You lose an electron, the ion is positive. How can adding something make something else negative and yet losing something makes it positive? Isn’t that backwards?
Well, those types of notation are due to human interpretation of nature. We call the pursuit of learning how all things work in the natural world science. What has been published in books is based on human interpretation of what has been tested, seen, or experienced through experimentation, observation, or mathematical analysis. +
This tiny eBook is about balancing equations because balanced equations are something scientists like to use. It reinforces the idea that we humans can’t spontaneously create matter or energy. All atoms come from someplace as does the energy used in all of our systems. The only way we can get “new” atoms are if they land on earth from outer space, or we create them in a huge machine. The atoms created in the huge machines are usually created just so we can see if that type of atom is able to exist. These newborns usually only last for fractions of a second.
As you will learn, if you have not studied them yet, molecules like to have their spatial arrangement balanced. They like symmetry and are more stable when the electrons are spaced out in a regular pattern. The idea of balance, equal spaces, equal quantities, and relationships between and among quantities permeates chemistry. With equations, you are using the language of chemistry to show the relationship between substances that are involved in a reaction.
To start with, a very simple scenario:
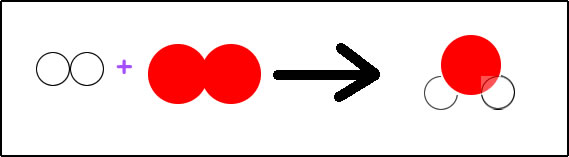
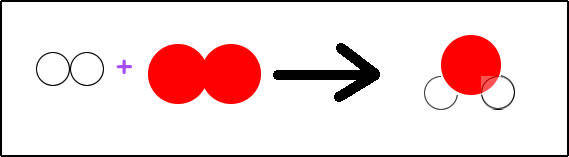
Hydrogen gas will combine with oxygen gas to form water.
Written with symbols: H2 + O2 --> H2O
Why would we want to balance equations? Is there really a point to it? Well, it turns out that nature likes things to be organized, even though things spontaneously fall apart. Chemistry is full of what often feels like contradictions. You add an electron, the ion becomes more negative. You lose an electron, the ion is positive. How can adding something make something else negative and yet losing something makes it positive? Isn’t that backwards?
Well, those types of notation are due to human interpretation of nature. We call the pursuit of learning how all things work in the natural world science. What has been published in books is based on human interpretation of what has been tested, seen, or experienced through experimentation, observation, or mathematical analysis. +
This tiny eBook is about balancing equations because balanced equations are something scientists like to use. It reinforces the idea that we humans can’t spontaneously create matter or energy. All atoms come from someplace as does the energy used in all of our systems. The only way we can get “new” atoms are if they land on earth from outer space, or we create them in a huge machine. The atoms created in the huge machines are usually created just so we can see if that type of atom is able to exist. These newborns usually only last for fractions of a second.
As you will learn, if you have not studied them yet, molecules like to have their spatial arrangement balanced. They like symmetry and are more stable when the electrons are spaced out in a regular pattern. The idea of balance, equal spaces, equal quantities, and relationships between and among quantities permeates chemistry. With equations, you are using the language of chemistry to show the relationship between substances that are involved in a reaction.
To start with, a very simple scenario:
Hydrogen gas will combine with oxygen gas to form water.
Written with symbols: H2 + O2 --> H2O

Hydrogen atoms do not occur by themselves in nature. It will always be attached to another atom. It is not stable being by itself. If the hydrogen is not forming a compound with another element, then it will occur in nature as H2, diatomic hydrogen.

The same thing is true for oxygen. Oxygen is most stable if it is attached to another atom. In nature, it is often attached to another oxygen atom, becoming diatomic oxygen, O2.

Now when hydrogen and oxygen combine, they form a new compound, dihydrogen monoxide. We call it water, H2O. Water, H2O, always occurs with hydrogen and oxygen atoms being in the same ratio. There are always two hydrogen atoms connected to one oxygen atom.
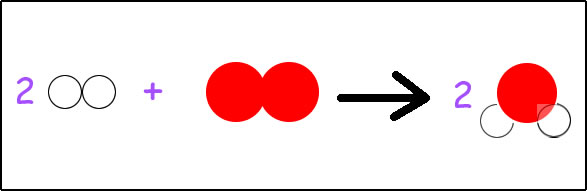
To show the reaction of the synthesis of water, we can just write H2 + O2 --> H2O. If, however, we want to do anything with the information in this equation, we need to balance it. According to this equation, hydrogen is available in sets of two atoms joined together. Oxygen is available as two oxygen atoms joined together. Water is available as two hydrogen atoms joined with one oxygen atom. Looking at the parts of the equation, we have on the left, two atoms of hydrogen and two atoms of oxygen. On the right side of the arrow, we have two atoms of hydrogen and one atom of oxygen. To be balanced, we need the same number of atoms on each type on both sides of the arrow. Are we currently balanced?
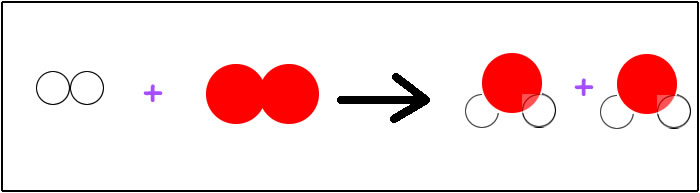
No, we are not. We have an extra oxygen on the left side. This means we need to add an oxygen to the right side, but oxygen can only exist on the right side it if is attached to two hydrogen atoms. Oxygen comes packaged as H2O. So… we will add another molecule of H2O to the right side. Our equation becomes: H2 + O2 --> H2O + H2O. Now we have two hydrogens on the left, two oxygens on the left, and on the right side we have four hydrogens and two oxygens. Are we balanced?
Once again, we are not. We now have two more hydrogen on the right side than the left side. How can we get more hydrogen on the left side? How do hydrogens come packaged? If you said we add one more molecule of diatomic H2 to the left side, you are correct! This will give us: H2 + H2 + O2 --> H2O + H2O.
Once again, we are not. We now have two more hydrogen on the right side than the left side. How can we get more hydrogen on the left side? How do hydrogens come packaged? If you said we add one more molecule of diatomic H2 to the left side, you are correct! This will give us: H2 + H2 + O2 --> H2O + H2O.
Now writing out the equation that way is technically correct. We are showing which molecules we need on each side for the equation to be balanced: the same number of each type of atom on both sides of the arrow. Writing out the long equation, however, takes more time and effort than writing it in a short-hand way, so we write it in short-hand. Just like in algebra x + x + x = 3x, H2 + H2 = 2H2. Similarly, like in algebra the 3 would be called a coefficient, the two before the H2 is also called a coefficient. The two written to the lower right is called a subscript.
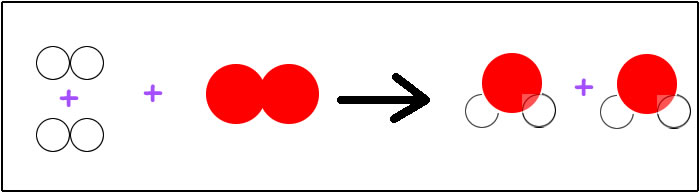
H2 + H2 + O2 --> H2O + H2O
H2 + H2 + O2 --> H2O + H2O
gets rewritten as: 2H2 + O2 --> 2H2O.
There are a few ways to interpret the coefficients. This can be read as two molecules of diatomic hydrogen plus one molecule of diatomic oxygen will produce two molecules of water. We could also say the mole ratio of hydrogen to oxygen is 2 to 1 or 2:1. If we wanted to confuse ourselves, we could say that three moles of reactants yields two moles of products. Since there is a direct relationship between the amount of gas at standard temperature and pressure (STP), we could say that at STP two liters of hydrogen plus one liter of oxygen will produce two liters of water vapor.
Student Assignment:
Students are to write out an explanation of how to balance a chemical equation. I have equations they can pick, and a sample of a book I wrote in the style I want them to do.
More details about the assignment are on this page: Student Assignment
The equation I'm showing how to balance:
3Zn(OH)2 + 2 H3PO4 --> Zn3(PO4)2 + 3 H2O
My example of the type of book I want students to make is embedded below.
Students are to write out an explanation of how to balance a chemical equation. I have equations they can pick, and a sample of a book I wrote in the style I want them to do.
More details about the assignment are on this page: Student Assignment
The equation I'm showing how to balance:
3Zn(OH)2 + 2 H3PO4 --> Zn3(PO4)2 + 3 H2O
My example of the type of book I want students to make is embedded below.
Equations they can balance:
20 Experience points level:
As + NaOH --> Na3AsO3 + H2
V2O5 + HCl --> VOCl3 + H2O
HClO4 + P4H10 --> H3PO4 + Cl2O7
Ca3(PO4)2 + H2SO4 --> CaSO4 + Ca(H2PO4)2
15 Experience points level:
Ba(NO3)2 + H3PO4 --> Ba3(PO4)2 + HNO3
AlCl3(aq) + NaOH(aq) --> NaAlO2(aq) + NaCl(aq) + H2O(l
Hg(OH)2 + H3PO4 --> Hg3(PO4)2 + H2O
H3PO4 + HCl --> PCl5 + H2O
10 Experience points level:
Au2S3 + H2 --> Au + H2S
Fe2O3 + H2 --> Fe + H2O
Mg + AgNO3 --> Mg(NO3)2 + Ag
SiO2 + HF --> SiF4 + H2O
N2 + O2 + H2O --> HNO3
CO + H2 --> C8H18 + H2O
H2SO4 + Pb(OH)4 --> Pb(SO4)2 + H2O
Na3PO4 + HCl --> NaCl + H3PO4
Ba3N2 + H2O --> Ba(OH)2 + NH3
5 Experience points level:
Mg(ClO3)2 --> MgCl2 + O2
AgNO3 + H2S --> Ag2S + HNO3
Al + N2 --> AlN
Zn + HCl --> ZnCl2 + H2
CH4 + O2 --> CO2 + H2O
Al + O2 --> Al2O3
Al +HCl --> AlCl3 + H2
AgI + Na2S --> Ag2S + NaI
TiCl4 + H2O --> TiO2 + HCl
20 Experience points level:
As + NaOH --> Na3AsO3 + H2
V2O5 + HCl --> VOCl3 + H2O
HClO4 + P4H10 --> H3PO4 + Cl2O7
Ca3(PO4)2 + H2SO4 --> CaSO4 + Ca(H2PO4)2
15 Experience points level:
Ba(NO3)2 + H3PO4 --> Ba3(PO4)2 + HNO3
AlCl3(aq) + NaOH(aq) --> NaAlO2(aq) + NaCl(aq) + H2O(l
Hg(OH)2 + H3PO4 --> Hg3(PO4)2 + H2O
H3PO4 + HCl --> PCl5 + H2O
10 Experience points level:
Au2S3 + H2 --> Au + H2S
Fe2O3 + H2 --> Fe + H2O
Mg + AgNO3 --> Mg(NO3)2 + Ag
SiO2 + HF --> SiF4 + H2O
N2 + O2 + H2O --> HNO3
CO + H2 --> C8H18 + H2O
H2SO4 + Pb(OH)4 --> Pb(SO4)2 + H2O
Na3PO4 + HCl --> NaCl + H3PO4
Ba3N2 + H2O --> Ba(OH)2 + NH3
5 Experience points level:
Mg(ClO3)2 --> MgCl2 + O2
AgNO3 + H2S --> Ag2S + HNO3
Al + N2 --> AlN
Zn + HCl --> ZnCl2 + H2
CH4 + O2 --> CO2 + H2O
Al + O2 --> Al2O3
Al +HCl --> AlCl3 + H2
AgI + Na2S --> Ag2S + NaI
TiCl4 + H2O --> TiO2 + HCl